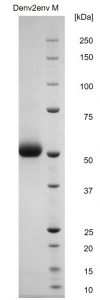
Reducing SDS-PAGE gel showing purified Dengue virus serotype 2 envelope protein running with an expected MW of approximately 50-55kDa.
Dengue Virus Serotype 2 Envelope Protein (HEK293)
We have replaced our Flavivirus envelope range with more effective VLPs and compact DIII antigens, ideal for multiplex and bead-based assays.
Contact us at NAC.Contact@LGCGroup.com for more information!
DENGUE VIRUS SEROTYPE 2 ENVELOPE PROTEIN (HEK293)
Dengue virus serotype 2 Envelope protein (E protein) has been manufactured in response to the unmet need for highly purified, concentrated protein for use in serological based diagnostic assays. Dengue virus serotype 2 Envelope protein is engineered in human cells using state-of-the-art expression and purification techniques. The soluble domain of the Dengue virus serotype 2 Envelope protein is expressed in HEK293 cells, and purified by IMAC and IEX chromatography.
PRODUCT DETAILS – DENGUE VIRUS SEROTYPE 2 ENVELOPE PROTEIN (HEK293)
- Recombinant Dengue virus serotype 2 Envelope protein (NCBI Accession Number: P14337.2).
- Manufactured in HEK293 cells with greater than 95% purity as determined by SDS-PAGE.
- Sequence contains amino acids 281-675 of the DENV2 Envelope protein, in addition to a C-terminal His-tag.
- Buffered in 20mM Tris-HCl, 110mM sodium chloride, pH7.8.
- Produced for Dengue virus immunoassay development.
BACKGROUND
Dengue virus is a member of the flavivirus family, which includes Zika virus, West Nile virus and Japanese Encephalitis virus. Many of the proteins expressed by these viruses are very similar, and serological testing for these viruses can be complicated by problems of cross-reactivity. In this context, the use of highly purified antigens, that are glycosylated and folded as native proteins can be highly important in the development of accurate immunoassays.
With more than one-third of the world’s population living in areas at risk of transmission, dengue infection is a leading cause of illness and death in the tropics and subtropics (ref WHO for Guidelines for Diagnosis, Treatment, Prevention & Control, 2009) As many as 100 million people are infected yearly.
Dengue is caused by any one of four related viruses transmitted by mosquitoes. It is a febrile illness that affects infants, young children and adults with symptoms appearing 3-14 days after the infective bite. Symptoms range from mild fever, to incapacitating high fever, with severe headache, pain behind the eyes, muscle and joint pain, and rash. There is no vaccine or any specific medicine to treat dengue. Severe dengue (also known as dengue hemorrhagic fever) is characterized by fever, abdominal pain, persistent vomiting, bleeding and breathing difficulty and is a potentially lethal complication, affecting mainly children. Early clinical diagnosis and careful clinical management by trained physicians and nurses increase survival of patient.
The first dengue vaccine, Dengvaxia (CYD-TDV) by Sanofi Pasteur, was first licensed in December, 2015, in Mexico. Several other vaccine candidates are in clinical or pre-clinical development