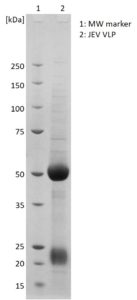
SDS-PAGE: Coomassie-stained SDS-PAGE showing purified JEV VLPs.
Japanese Encephalitis Virus VLP
Price range: £400.07 through £1,700.31 excl. VAT
Recombinant JEV virus-like particles (sequence strain SA-14) composed of pre-Membrane and Envelope proteins expressed in HEK293 cells and purified from culture supernatant using density gradient centrifugation and ion exchange chromatography.
JAPANESE ENCEPHALITIS VIRUS VLP
Recombinant Japanese encephalitis VLP (virus-like particles) produced in HEK293 cells and purified from culture supernatant.
PRODUCT DETAILS – JAPANESE ENCEPHALITIS VIRUS VLP
- VLPs composed of pre-Membrane and Envelope proteins (NCBI accession number P27395.1, AA105-819; sequence strain SA-14).
- Expressed in HEK293 cells and purified from culture supernatant using density gradient centrifugation and ion exchange chromatography.
- Presented in Dulbecco’s phosphate buffered saline pH7.4, sterile filtered.
BACKGROUND
The Japanese encephalitis virus (JEV) is a flavivirus and is related to the West Nile Virus. JEV is the main cause of viral encephalitis in many countries of Asia with an estimated 68 000 clinical cases every year (WHO). Although symptomatic Japanese encephalitis (JE) is rare, the case-fatality rate among those with encephalitis can be as high as 30%. Permanent neurologic or psychiatric sequelae can occur in 30%–50% of those with encephalitis. Flaviviruses are positive-stranded RNA viruses with a 9-12- kb genome that is translated as a single polyprotein and then cleaved by host and viral proteases into structural capsid (C), premembrane (prM), envelope (E) proteins and seven nonstructural proteins. Capsid binds to viral RNA to form a nucleocapsid that is enveloped by an endoplasmic reticulum-derived membrane containing E and prM. E proteins are responsible for cellular attachment and possess a hydrophobic loop that mediates fusion of viral and host membrane.The E protein is therefore the primary target of neutralizing antibodies.
VLPs are often spontaneously produced during Flavivirus infection and they can also be produced recombinantly using different expression systems (Kümmerer, 2006). VLPs are produced as a noninfectious recombinant antigens and expressed as extracellular particles. Studies have showm that gradient-purified JEV particles exhibit hemagglutination activity at the same pH optimum (6.6) as Japanese encephalitis virus. IgM and IgG ELISA have also shown that recombinantly produced JEV VLPs perform similarly to Japanese encephalitis virus-infected mouse brain antigen for the analysis of serum samples from patients with symptoms of acute encephalitis (Hunt et al. 2001).
Many diagnostic reagents and vaccines targetting viral diseases are produced using infectious agents, making them costly and hazardous to manufacture. However, recombinant DNA technology, makes it possible to bypass these problems by synthesizing viral immunogens and antigens in vitro. To be viable, the immunologically relevant viral components must be produced in an authentic form, which often require the recombinant expression system to duplicate the posttranslational processing pathways that contribute to viral antigen formation. These posttranslational events in antigen formation may be particularly important in the synthesis of envelope glycoprotein structures, especially those that are heterodimeric (Konishi et al., 2001). This Japanese Encephalitis Virus VLP has been manufactured using mammalian cells to meet the need for a highly purified, concentrated protein for use in vaccine research and serological based diagnostic products (Krol et al., 2019). As usual this product is produced to The Native Antigen Company’s very high quality standards for lot-to-lot variation minimisation and product traceability. Please see the downloadable Certificate of Analysis for further information.
REFERENCES
- Hunt et al. (2001). A recombinant particulate antigen of Japanese encephalitis virus produced in stably-transformed cells is an effective noninfectious antigen and subunit immunogen. Journal of Virological Methods. Volume 97, Issues 1–2, Pages 133-149.
- Japanese encephalitis. 2019. World Health Organisation (WHO).
- Konishi et al. (2001). Generation and Characterization of a Mammalian Cell Line Continuously Expressing Japanese Encephalitis Virus Subviral Particles. J Virol. 75(5): 2204–2212.
- Krol et al. (2019). Production and Biomedical Application of Flavivirus-like Particles. Trends in Biotechnology.
- Kümmerer B. The molecular biology of yellow fever virus. in: Kalitzky M. Borowski P. Molecular Biology of the Flavivirus. Horizon Bioscience, 2006: 1-16.