Human anti-Varicella zoster virus (VZV) gH IgM (clone M1484), Recombinant
Price range: $674.73 through $2,867.60 excl. VAT
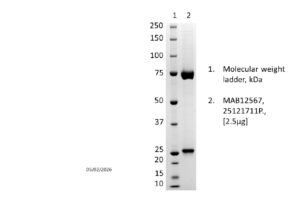
Recombinant human anti-Varicella zoster virus VZV gH (clone M1484) IgM antibody, purified by ion exchange chromatography, ammonium sulphate precipitation and dialysis.
Human anti-Varicella zoster virus (VZV) gH IgM (clone M1484), Recombinant
Recombinant human anti-Varicella zoster virus VZV gH (clone M1484) IgM antibody, purified by ion exchange chromatography, ammonium sulphate precipitation and dialysis..
PRODUCT DETAILS – Human anti-Varicella zoster virus (VZV) gH IgM, Recombinant
- Isotype: human IgM
- Clone Number: M1484
- Presented as Liquid in 50mM HEPES, 150mM NaCl, pH8.0
BACKGROUND
Varicella‑zoster virus (VZV) is a strictly human, neurotropic alphaherpesvirus responsible for varicella (chickenpox) upon primary infection and herpes zoster (shingles) upon reactivation from latency within sensory ganglia, with the disease burden primarily affecting older adults and immunocompromised individuals. The viral envelope carries several surface glycoproteins—most notably glycoproteins E (gE), B (gB), and H/L (gH/gL)—which serve as major targets of the humoral immune response and are essential for virion fusion, cellular entry, and direct cell‑to‑cell transmission (Sullivan et al., 2018). Experimental studies employing monoclonal antibodies indicate that gH/gL and gB are the minimal components required for VZV-mediated membrane fusion, and that anti‑gH monoclonal antibodies effectively neutralize viral particles, suppress intercellular spread, and influence glycoprotein localization within infected cells (Shiraki et al., 2011).
Recombinant VZV-specific antibodies offer a renewable, precisely defined alternative to traditional hybridoma-derived antibodies for probing these glycoprotein-directed immune responses (Birlea et al., 2013; Sullivan et al., 2018). Human recombinant monoclonal antibodies isolated following Zostavax immunization have been shown to bind conformational epitopes within the gH/gL complex and neutralize VZV infection in vitro (Birlea et al., 2013), facilitating fine‑scale studies of viral membrane fusion and epitope recognition. In combination with recombinant gE‑based assays, such as indirect ELISAs for detecting anti‑VZV antibodies in clinical specimens (Niu et al., 2022), these reagents form the foundation of contemporary serological testing, vaccine performance assessment, and the rational design of antibodies with enhanced specificity and functional properties for diagnostic and potential therapeutic use (Sullivan et al., 2018).
REFERENCES
Birlea M et al. Human anti varicella zoster virus (VZV) recombinant monoclonal antibody produced after Zostavax immunization recognizes the gH/gL complex and neutralizes VZV infection. J Virol. 2013
Sullivan NL et al. Breadth and functionality of varicella zoster virus glycoprotein specific antibodies identified after Zostavax vaccination in humans. J Virol. 2018.
Shiraki K et al. Neutralizing anti gH antibody of varicella zoster virus modulates distribution of gH and induces gene regulation, mimicking latency. J Virol. 2011.
Niu Y et al. Development of an indirect ELISA kit for rapid detection of varicella zoster virus antibody by glycoprotein E. Front Microbiol. 2022.
You may also like…
Varicella-Zoster Virus Heterodimer gE/gI (HEK293), His-tag
Price range: $512.90 through $1,801.46 excl. VAT
Human anti-Varicella zoster virus (VZV) gE IgM, Recombinant
Price range: $674.73 through $2,867.60 excl. VAT
Human anti-Varicella zoster virus (VZV) gH IgM (clone M1416), Recombinant
Price range: $674.73 through $2,867.60 excl. VAT