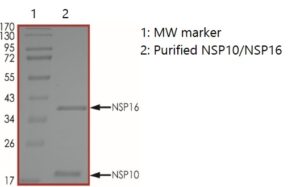
SDS-PAGE: Coomassie-stained reducing SDS-PAGE showing purified NSP10/NSP16 methyltransferase.
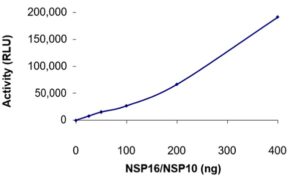
ELISA: The specific activity of SARS-CoV-2 NSP10/NSP16 methyltransferase was determined to be 53 pmol/min/mg (Methyltransferase-Glo™ Methyltransferase Assay, Promega).
SARS-CoV-2 NSP10/NSP16 Methyltransferase, Active
Price range: £417.70 through £816.41 excl. VAT
SARS-CoV-2 NSP10/NSP16 methyltransferase is an active recombinant protein expressed in E. coli using a C-terminal His tag. SARS-CoV-2, previously known as the 2019 Novel Coronavirus (2019-nCoV), causes the pandemic COVID-19 disease.
SARS-COV-2 NSP10/NSP16 METHYLTRANSFERASE, ACTIVE
SARS-CoV-2 NSP10/NSP16 methyltransferase is a full-length active recombinant protein expressed in E. coli using a C-terminal His tag. SARS-CoV-2, previously known as the 2019 Novel Coronavirus (2019-nCoV), causes the pandemic COVID-19 disease.
PRODUCT DETAILS – SARS-COV-2 NSP10/NSP16 METHYLTRANSFERASE, ACTIVE
- SARS-CoV-2 NSP10/NSP16 methyltransferase, full-length.
- Expressed in E. coli cells with an C-terminal His-tag and >90% purity.
- Presented as liquid in 50mM sodium phosphate, pH 7.0, 300mM NaCl, 150mM imidazole, 0.25mM DTT, 25% glycerol.
- Specific activity is 53 pmol/min/mg (Methyltransferase-Glo™ Methyltransferase Assay, Promega).
BACKGROUND
In December 2019 a novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), formerly known as the 2019 novel coronavirus (2019-nCoV) was identified in Wuhan, China, causing a world-wide pandemic (Wu et al., 2020). Three coronaviruses, SARS-CoV, MERS-CoV, and SARS-CoV-2 have been identified as being a highly pathogenic for humans, and there is currently no effective antiviral treatment. Therefore, studies are focused on rapid development of vaccines and antiviral drugs to prevent and treat coronavirus infection. There are several potential strategies to pharmacologically fight against the disease (COVID-19), including vaccines, monoclonal antibodies, oligonucleotide-based therapies, peptides, interferon therapies, and small-molecule drugs (Dömling & Gao, 2020).
Many eukaryotic viruses have evolved 2′-O-methyltransferases (2′-O-MTase) to modify their viral mRNAs and carry a cap-1 structure (m7GpppNm) at the 5′ end. This 5’ cap structure is important for viral mRNA stability, protein translation and viral immune escape (Furuichi et al., 2000). SARS-CoV possess NSP16 that has 2′-O-MTase activity. NSP16 requires NSP 10 for activation which is a conserved mechanism in corona viruses (Decroly et al., 2008). Mutations have been identified in SARS-CoV-2 NSP10 and NSP16, which alter NSP10-NSP-16 secondary structure, and protein-modelling studies have revealed that such mutations can affect the proteins dynamicity and stability (Azad et al., 2020). Therefore, inhibitors targeting the NSP10/NSP16 2′-O-MTase are potential targets for developing anti-coronavirus drugs (Chen et al., 2011).
REFERENCES
- Azad GK. Identification of novel mutations in the methyltransferase complex (Nsp10-Nsp16) of SARS-CoV-2. Biochem Biophys Rep. 2020 Dec;24:100833.
- Chen, Y. et al: Biochemical and structural insights into the mechanisms of SARS coronavirus RNA ribose 2’-O-methylation by nsp16/nsp10 protein complex. PLoS Pathog. 2011, 7(10): e1002294.
- Decroly, E. et al. Coronavirus nonstructural protein 16 is a cap-o binding enzyme possessing (nucleoside-2’O)-methyltransferase activity. J Virol. 2008, 82(16):8071-8084.
- Dömling A, Gao L. Chemistry and Biology of SARS-CoV-2. Chem. 2020;6(6):1283-1295.
- Furuichi, Y. et al: Viral and cellular mRNA capping: past and prospects. Adv Virus Res. 2000, 55:135–184.