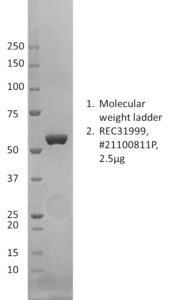
SDS-PAGE: Representative Coomassie-stained reducing SDS-PAGE showing purified nucleoprotein.
Influenza A [A/Hong Kong/1/1968 (H3N2)], Nucleoprotein (NP), His-Tag
£330.90 - £1,257.16
£264.72 - £1,005.73
excl. VAT
Influenza A [A/Hong Kong/1/1968 (H3N2)] nucleoprotein (NP) is a recombinant antigen expressed and purified from Escherichia coli.
INFLUENZA A (A/HONG KONG/1/1968 (H3N2)] NUCLEOPROTEIN (NP), HIS-TAG
Influenza A [A/Hong Kong/1/1968 (H3N2)] nucleoprotein (NP) is a recombinant antigen expressed and purified from Escherichia coli.
PRODUCT DETAILS – INFLUENZA A (A/HONG KONG/1/1968 (H3N2)] NUCLEOPROTEIN (NP), HIS-TAG
- Influenza A [A/Hong Kong/1/1968 (H3N2)] nucleoprotein (NP)
- Protein is expressed and purified from Escherichia coli to >95% purity
- Presented in 50mM Tris-HCl, pH 8.0, 400mM NaCl.
BACKGROUND
Of the three types of influenza virus (A, B and C) A is the best characterized and the most serious threat to public health. One of the three core Influenza polypeptides is an RNA-dependent RNA polymerase and a single-strand RNA (ssRNA) binding protein, often referred to as a nucleoprotein (NP). NP encapsidates the virus genome to form a ribonucleoprotein (RNP) particle for the purposes of transcription and packaging. NP is not just the structural component of the virus transcription machinery but performs multiple essential functions throughout the virus life cycle (Portela & Digard, 2002). Influenza A virus RNA segment 5 encodes NP is a polypeptide of 498 amino acids in length; in influenza B and C viruses, the length of the homologous NP polypeptide is 560 and 565 residues, respectively (Londo et al., 1983; Nakada et al., 1984). Alignment of the predicted amino acid sequences of the NP genes of the three influenza virus types reveals significant similarity among the three proteins, with the type A and B NPs showing the highest degree of conservation. Phylogenetic analysis of virus strains isolated from different hosts reveals that the NP gene is also well conserved, with a maximum amino acid difference of less than 11% (Shu et al., 1993). The primary sequence of NP is modified by phosphorylation (Privalsky & Penhoet, 1978) and the phosphopeptide pattern of NP is strain-specific, dependent on the host cell used for propagating the virus and changes throughout virus replication (Kistner et al., 1985, 1989). Seasonal influenza viruses display high variability and because of the threat of influenza viruses with pandemic potential, there is great interest in the development of vaccines that induce broadly protective immunity, such as those based on conserved proteins, including nucleoprotein. NP has also been shown to induce cross-reactive antibody and T cell responses (Altenburg et al., 2017).
REFERENCES
- Altenburg AF, Magnusson SE, Bosman F, Stertman L, de Vries RD, Rimmelzwaan GF. Protein and modified vaccinia virus Ankara-based influenza virus nucleoprotein vaccines are differentially immunogenic in BALB/c mice. Clin Exp Immunol. 2017 Oct;190(1):19-28.
- Londo DR, Davis AR, Nayak DP. Complete nucleotide sequence of the nucleoprotein gene of influenza B virus. J Virol. 1983 Sep;47(3):642-8.
- Kistner O, Müller H, Becht H, Scholtissek C. Phosphopeptide fingerprints of nucleoproteins of various influenza A virus strains grown in different host cells. J Gen Virol. 1985 Mar;66 ( Pt 3):465-72.
- Kistner O, Müller K, Scholtissek C. Differential phosphorylation of the nucleoprotein of influenza A viruses. J Gen Virol. 1989 Sep;70 ( Pt 9):2421-31.
- Nakada S, Creager RS, Krystal M, Palese P. Complete nucleotide sequence of the influenza C/California/78 virus nucleoprotein gene. Virus Res. 1984 Sep;1(6):433-41.
- Portela A, Digard P. The influenza virus nucleoprotein: a multifunctional RNA-binding protein pivotal to virus replication. J Gen Virol. 2002 Apr;83(Pt 4):723-734.
- Shu LL, Bean WJ, Webster RG. Analysis of the evolution and variation of the human influenza A virus nucleoprotein gene from 1933 to 1990. J Virol. 1993 May;67(5):2723-9.


![Influenza A [A/Hong Kong/483/97 (H5N1)] Hemagglutinin (HA), His-Tag](https://thenativeantigencompany.com/wp-content/uploads/1970/01/shutterstock_1204546204-300x300.jpg)
![Influenza A [A/Hong Kong/45/2019 (H3N2)]](https://thenativeantigencompany.com/wp-content/uploads/2020/12/Misc-Virus-122-300x300.jpg)