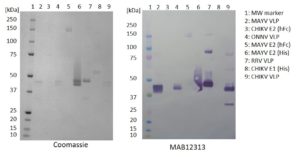
Western blot (non-reducing): 100ng of each antigen was used for SDS-PAGE, in non-reduced form. Proteins were transferred using Transblot for 7 min. at 25V. 5% dry milk in PBS-T was used as blocking buffer and dilution buffer for antibodies. Primary antibody (MAB12313) and goat anti-mouse-IgG-HRP secondary antibody (Biorad 103005) were used at 1:1000. All steps were carried out for 1h at room temperature with gentle rocking. KPL Membrane TMB was used for detection. Development time 30 sec.
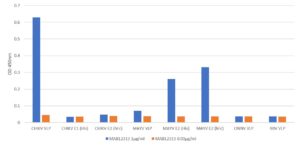
ELISA: Plate was coated with 0.5µg/ml of antigens in 1X DPBS overnight at 2-8°C. Blocked for 1.5h using 1%BSA/DPBS.300µl/well. Washed plate 3X using Tris wash buffer. Added 100µl of prepared antibody dilutions. Incubated for 2h at RT at 800rpm.Washed plate 3X using Tris wash buffer. Added 100µl of Goat anti mouse IgG HRP antibody (1:5000 dilution) to all the wells and incubated for 1h at RT, 800rpm. Washed plate 4X using Tris wash buffer. Added 100µl HKTMB and incubated for ~2min. at RT. Added 100µl 1M HCl to stop the reaction and read the plate at 450nm.
MOUSE ANTI-CHIKUNGUNYA VIRUS VLP (AG7)
This mouse anti chikungunya virus VLP antibody recognises Chikungunya virus VLPs containing E1, E2 and capsid proteins.
PRODUCT DETAILS – MOUSE ANTI-CHIKUNGUNYA VIRUS VLP (AG7)
- Mouse anti Chikungunya VLP antibody recognises the Chikungunya virus VLP, containing E1, E2 and Capsid proteins.
- Immugen comprised an equal mix of Chikungunya, Mayaro and O’nyong’nyong virus VLPs (E1, E2 and Capsid proteins).
- Antibody shows some cross-reactivity with Mayaro Envelope protein in ELISA but not with O’nyong’nyong virus or Ross River virus VLPs.
- Antibody recognises VLPs for CHIKV, ONNV, MAYV and RRV in non-reducing Western blot. Antibody not suitable for Western blot in reducing conditions.
BACKGROUND
Chikungunya virus is a member of the genus Alphavirus in the family Togaviridae. Chikungunya fever is a mosquito-borne disease first identified in Tanzania in 1953. Since 2004 there have been extensive outbreaks in Africa and Asia, and in 2013 the first cases were identified in the Caribbean, and by September 2014 more than 650,000 cases had been reported in the Americas. Chikungunya fever usually starts 2–4 days after chikungunya virus infection, with clinical symptoms including high fever, rash, headache, vomiting, myalgia, and severe joint pain.
Diagnosis of CHIKV may be hampered by the fact that clinical symptoms of CHIKV are similar to those seen in cases of Dengue Virus and Zika Virus infection. Therefore, differential diagnosis is an important consideration in areas where flaviviruses such as Dengue and Zika co-circulate. Currently, there is no specific treatment available for the treatment of Chikungunya fever or licensed vaccine for the prevention of CHIKV infection.
Virus-Like Particles (VLPs) are an emerging vaccine technology and consist of protein shells comprising outer proteins specific to the virus in question. The Native Antigen Company’s Chikungunya virus VLP is composed of E1, E2 and capsid proteins, from Chikungunya virus sequences. Concentration and purification is then performed by a series of ultracentrifugation and chromatographical methods which result in VLPs of exceptional quality and purity. These antigens are then used to produce antibodies with higher affinity and greater sensitivity, which can used for multiple applications.