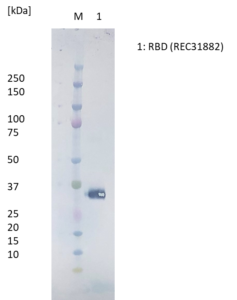
 Western Blot: RBD protein (50ng) loaded onto Novex 4-12% Bis-Tris gel, alongside Kaleidoscope marker (BioRad), and separated at 200V for 50 min. Polyclonal antibody added at 1mg/ml (1:1000) and secondary antibody (STAR124P) added at 1mg/ml (1:1000).
Western Blot: RBD protein (50ng) loaded onto Novex 4-12% Bis-Tris gel, alongside Kaleidoscope marker (BioRad), and separated at 200V for 50 min. Polyclonal antibody added at 1mg/ml (1:1000) and secondary antibody (STAR124P) added at 1mg/ml (1:1000).
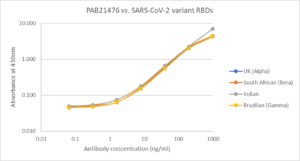
Direct ELISA: Plate was coated with 100µl of variant RBD proteins (UK, REC31946; South African, REC31945; Indian, REC31971; Brazilian, REC31961), at 1µg/ml and then incubated with 100µl PAB21476 antibody, diluted from 1000ng/ml to 0.064ng/ml. Diluted secondary IgG HRP antibody (100µl at 1:10,000) was then added. 100µl of TMB substrate (M0701A) was added in all wells and the reaction stopped after 10 min. with 1M HCl and the plate read at 405/450nm.
Rabbit IgG Anti SARS-CoV-2 Spike (S1) RBD Polyclonal Antibody
Price range: £431.26 through £1,338.53 excl. VAT
Rabbit anti SARS-CoV-2 (Covid-19) spike receptor binding domain (RBD) antibody, for use in ELISA and Western blot applications. Antibody recognizes RBD from Wuhan-Hu-1, UK (Alpha), South African (Beta) Brazilian (Gamma) and Indian variants. Antibody does not cross-react in ELISA with HCoV-229E full-length spike protein (REC31880). SARS-CoV-2, previously known as the 2019 Novel Coronavirus (2019-nCoV), causes the pandemic COVID-19 disease.
RABBIT IGG ANTI-SARS-COV-2 SPIKE (S1) RBD POLYCLONAL ANTIBODY
Rabbit anti SARS-CoV-2 (Covid-19) spike receptor binding domain (RBD) antibody, for use in ELISA and Western blot applications. Antibody recognizes RBD from Wuhan-Hu-1, UK (Alpha), South African (Beta) Brazilian (Gamma) and Indian variants. Antibody does not cross-react in ELISA with HCoV-229E full-length spike protein (REC31880).
PRODUCT DETAILS – RABBIT IGG ANTI-SARS-COV-2 SPIKE (S1) RBD POLYCLONAL ANTIBODY
- Anti SARS-CoV-2 RBD polyclonal antibody against Wuhan-Hu-1, UK (Alpha), South African (Beta) Brazilian (Gamma) and Indian variants.
- Antibody was prepared from whole rabbit serum produced by immunization with a purified recombinant SARS-CoV-2 RBD protein (REC31882).
- The product was purified from antiserum by protein A affinity purification.
- Antibody is presented in PBS with 0.09% sodium azide.
BACKGROUND
Severe acute respiratory syndrome (SARS) is a lower respiratory tract illness that was first reported in patients from the Guandong Province of China in November 2002. The causative agent, which was previously unknown, was isolated in 2003 and named as SARS coronavirus (SARS-CoV). The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the causative agent of the coronavirus induced disease 19 (COVID-19) which emerged in China in late 2019, resulting in a worldwide epidemic (Zhou et al., 2020).
Human coronaviruses are the major cause of upper respiratory tract illness. They are positive-stranded RNA viruses, and contain the largest viral RNA genomes known to date (27-31 kb). SARS (severe acute respiratory syndrome) and COVID-19 are both caused by human coronaviruses, SARS-CoV and SARS-CoV-2, respectively. The genome of SARS-CoV-2 shares 82% nucleotide identity with human SARS-CoV and 89% with bat SARS-like-CoVZXC21 (Lu et al., 2020; Zhao et al., 2020). However, initially it displayed lower pathogenicity and higher human to human transmissibility (Li et al., 2020). The coronavirus genome encodes four structural proteins: the spike (S) protein, nucleocapsid (N) protein, membrane (M) protein, and the envelope (E) protein. The S protein assists in the attachment of the virus to the human cell and comprises intracellular, transmembrane, and extracellular regions. The extracellular region contains the S1 receptor binding subunit (RBD) and the S2 membrane fusion subunit. Generally, following SARS-CoV-2 infection, antibodies appear after 7–14 days and persist for weeks after viral clearance. The most commonly detected antibodies are against the N protein and the S protein. Coronavirus neutralizing antibodies primarily target the trimeric spike (S) glycoproteins on the viral surface (Wang et al., 2020) and can change the course of infection in an infected individual by supporting virus clearance or protecting an uninfected host that is exposed to the virus (Prabakaran et al., 2009). However, the antibody responses against SARS-CoV-2 remain poorly understood (Tang et al., 2020) and better understanding of how the viral coating triggers a healthy immune system’s recognition and neutralisation of the virus is critical for optimisation of diagnostic tests (Petherick, 2020). It has been suggested that spike RBD may be a better target than N for diagnostic tests (Rosadas et al., 2020). The Native Antigens monoclonal antibodies, which are specific for SARS-CoV-2, have been manufactured to meet the need for improved COVID-19 diagnostic assays.
REFERENCES
- Lu, R., Zhao, X., Li, J., Niu, P., Yang, B., Wu, H., Wang, W., Song, H., Huang, B., Zhu, N., et al. (2020). Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 395, 565–574.
- Petherick A. Developing antibody tests for SARS-CoV-2. Lancet. 2020 Apr 4;395(10230):1101-1102.
- Prabakaran P, Zhu Z, Xiao X, Biragyn A, Dimitrov AS, Broder CC, Dimitrov DS. Potent human monoclonal antibodies against SARS CoV, Nipah and Hendra viruses. Expert Opin Biol Ther. 2009 Mar;9(3):355-68.
- Rosadas C, Randell P, Khan M, McClure MO, Tedder RS. Testing for responses to the wrong SARS-CoV-2 antigen? Lancet. 2020 Sep 5;396(10252):e23.
- Tang YW, Schmitz JE, Persing DH, Stratton CW. Laboratory Diagnosis of COVID-19: Current Issues and Challenges. J Clin Microbiol. 2020 May 26;58(6):e00512-20.
- Wang C, Li W, Drabek D, Okba NMA, van Haperen R, Osterhaus ADME, van Kuppeveld FJM, Haagmans BL, Grosveld F, Bosch BJ. A human monoclonal antibody blocking SARS-CoV-2 infection. Nat Commun. 2020 May 4;11(1):2251.
- Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020 Mar;579(7798):270-273.