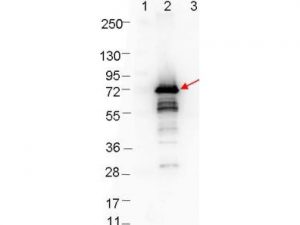
RABBIT ANTI-BORRELIA BURGDORFERI SENSU STRICTO (B31) OSPA ANTIBODY
Rabbit Anti-Borrelia burgdorferi OspA antibody, is a polyclonal suitable for use in ELISA and western blotting applications. Strain B31 is the type strain (ATCC 35210) for this organism and was derived by limited dilutional cloning from the original Lyme-disease tick isolate obtained by A. Barbour (Johnson, et al., 1984).
PRODUCT DETAILS – RABBIT ANTI-BORRELIA BURGDORFERI SENSU STRICTO (B31) OSPA ANTIBODY
- Rabbit anti-B. burgdorferi sensu stricto OspA polyclonal IgG antibody (strain B31).
- Greater than 95% purity by SDS-PAGE and buffered in 0.02 M Potassium Phosphate, 0.15 M Sodium Chloride, pH 7.2.
BACKGROUND
Outer-Surface Protein A (OspA) is a 31 kDa lipoprotein encoded by Borrelia burgdorferi and is a major component of the spirochete’s extracellular matrix (Stevenson, et al., 1996), probably associated with lipid rafts and serving as a lipid-anchor (Toledo, et al., 2014). Strain B31 is the type strain (ATCC 35210) for this organism and was derived by limited dilutional cloning from the original Lyme-disease tick isolate obtained by A. Barbour (Johnson, et al., 1984). The Borrelia species causing Lyme disease express different OspA serotypes on their surface, B. burgdorferi (serotype 1), B. afzelii (serotype 2), B. garinii (serotypes, 3, 5 and 6) and B. bavariensis (serotype 4) (Wilske, et al., 1988).
Many of the borrelial surface antigens are lipid-modified proteins (i.e. lipoproteins), although a number of these surface-exposed lipoproteins (OspA, OspB, and OspC) are not found exclusively on the surface of the organism. These lipoproteins are also detected in the periplasm of the organism and can be shuttled to and from the borrelial surface at different points during infection. The interface between B. burgdorferi and its human host is its outer surface (and therefore proteins localized to the outer membrane) play an important role in dissemination, virulence, tissue tropism, and immune evasion. Antibodies directed against outer surface proteins have also been shown to protect animals and humans from infection with B. burgdorferi (Kenedy, et al., 2012).
OspA likely mediates the attachment of B. burgdorferi to the tick mid-gut by binding the mid-gut receptor TROSPA (Tick Receptor for OspA). TROSPA is downregulated to allow migration out of the tick mid-gut during feeding, and into the salivary glands before being transmitted to the mammalian host. This transition is believed to be facilitated by changes in expression of some B. burgdorferi genes, including OspA (Ding, et al., 2000). These changes may be regulated by changes in tick life cycle, changes in conditions during tick feeding (such as temperature, pH and nutrients) and/or in coordination with the course of infection in the mammal host (Norris, 2006).
A heterodimer of the linked C-terminal half of two OspA serotypes was shown to protect mice from a challenge with spirochetes expressing either OspA serotype 1, 2 or 5, when challenged with infected ticks and in vitro grown spirochetes (Comstedt, et al., 2014). OspA-specific human Mabs can prevent the transmission of B. burgdorferi from ticks to mice (Wang, et al., 2016). More recently, structural analysis of the human antibody LA-2/OspA complex has revealed specific residues that may be exploited to modulate recognition of the protective epitope of OspA potentially offering a new path towards prophylactic passive antibodies (Shivender, et al., 2017).
REFERENCES
- Comstedt, P. et al., 2014. Design and Development of a Novel Vaccine for Protection against Lyme Borreliosis. PLoS One, 9(11), pp. 1-12.
- Ding, W. et al., 2000. Structural identification of a key protective B-cell epitope in Lyme disease antigen OspA. J. Mol. Biol. , Volume 302, pp. 1153-1164.
- Johnson, R.C., et al. 1984. Borrelia burgdorferi sp. nov.: etiologic agent of Lyme disease. Int J Syst Bacteriol, 34, pp. 496–497.
- Kenedy, M. R., Lenhart, T. R. & Akins, D. R., 2012. The Role of Borrelia burgdorferi Outer Surface Proteins. FEMS Immunol Med Microbiol., 66(1), pp. 1-19.
- Norris, S. J., 2006. The dynamic proteome of Lyme disease Borrelia. Genome Biol., 7(3), p. 209.
- Shivender, S. et al., 2017. Structural and Molecular Analysis of a Protective Epitope of Lyme Disease Antigen OspA and Antibody Interactions. J Mol Recognit., 30(5), pp. 1-15.
- Stevenson, B., Tilly, K. & Rosa, P. A., 1996. A Family of Genes Located on Four Separate 32-Kilobase Circular Plasmids in Borrelia burgdorferi B31. J Bacteriol, 178(12), pp. 3508-3516.
- Toledo, A. et al., 2014. Selective Association of Outer Surface Lipoproteins with the Lipid Rafts of Borrelia burgdorferi. MbIO, 5(2).
- Wang, Y. et al., 2016. Pre-exposure Prophylaxis With OspA-Specific Human Monoclonal Antibodies Protects Mice Against Tick Transmission of Lyme Disease Spirochetes. J Infect Dis., 214(2), p. 205–211.
- Wilske, B., 1988. Antigenic variability of Borrelia burgdorferi. Ann N Y Acad Sci, 539, pp. 126-43.