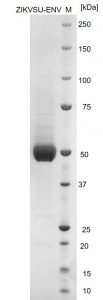
SDS-PAGE: Reducing SDS-PAGE gel showing purified Zika virus Envelope protein, showing protein migrating as a band of approximately 45-50kDa.
Zika Virus Envelope Protein (Suriname strain)
$1,124.40 – $4,808.94 excl. VAT
Recombinant Zika virus envelope protein expressed in HEK293 mammalian cells, with C terminal 6-His tag.
ZIKA VIRUS ENVELOPE PROTEIN
Zika virus Envelope protein (E protein) has been manufactured in response to the unmet need for highly purified, concentrated antigen for use in serological based diagnostic assays. Recombinant Zika virus Envelope protein is engineered in human cells using state-of-the-art expression and purification techniques. The soluble domain of the Zika virus Envelope is expressed in HEK293 cells, and purified by IMAC and IEX chromatography.
PRODUCT DETAILS – ZIKA VIRUS ENVELOPE PROTEIN
- Recombinant Zika virus Envelope protein (strain Suriname Z1106033, NCBI Accession Number: AVZ25034.1).
- Includes amino acids 291-696 and a C-terminal His-tag.
- Greater than 95% purity by SDS-PAGE and buffered in 20mM Tris-HCl, 220mM NaCl pH7.8.
BACKGROUND
Zika virus (ZIKV) is a member of the Flavivirus genus of the family Flaviviridae that also includes global human pathogenic viruses Dengue virus (DENV), Yellow Fever virus, West Nile virus and Japanese encephalitis virus. Zika infection is an emerging disease caused by Zika virus, which is spread through the bite of infected Aedes mosquitoes. The Zika virus was first isolated in Central Africa but has since spread to South Asia and South America. The Zika virus can also be transmitted to individuals through sexual contact, blood product transfusion and organ transplant (WHO, 2018).Zika virus can also be transmitted to the foetus during pregnancy causing microcephaly (CDC).
Healthy individuals infected with Zika virus may remain asymptomatic or can experience mild fever, rash, myalgia, arthralgia and headaches. In addition, Zika virus infection in adults has been associated with neuropathy, myelitis and the neurological condition Guillain-Barre syndrome. Due to similar symptoms, Zika virus infection can be misdiagnosed as a Dengue virus infection and vice-versa. Currently, there are no vaccines available for the prevention of Zika virus. Therefore, accurate diagnosis of ZIKV is essential for patient management especially in regions where DENV and ZIKV co-circulate. However, ZIKV and DENV show structural and genetic similarities, and serological testing for the viruses can be complicated by problems of cross-reactivity.
REFERENCES
- Zika Virus Factsheet. World Health Organization (WHO), 2018.
- Centers for Disease Control and Prevention. CDC Concludes Zika Causes Microcephaly and Other Birth Defects.
Publishing research using our reagents? Please let us know so that we can cite your publication as a reference.
- Wen J et al (2017). Dengue virus-reactive CD8+ T cells mediate cross-protection against subsequent Zika virus challenge. Nat Commun. Nov 13;8(1):1459. PMID: 29129917