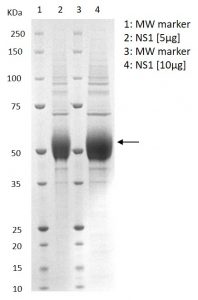
SDS-PAGE showing purified recombinant NS1 protein.
Saint Louis Encephalitis Virus NS1 Protein
£450.24 - £1,607.05 excl. VAT
Recombinant Saint Louis encephalitis virus NS1 protein produced in HEK293 cells, incorporating a C-terminal 6x His-tag.
Note: Discounts shown below are for the smallest vial size.
| Quantity | Discount | Price |
|---|---|---|
| 5-9 | 15% | £382.70 |
| 10-Unlimited | 30% | £315.17 |
SAINT LOUIS ENCEPHALITIS VIRUS NS1 PROTEIN
This is a recombinant Saint Louis encephalitis virus NS1 protein produced in HEK293 cells. The protein contains a C-terminal 6x His-tag. Saint Louis encephalitis is a disease caused by the mosquito borne Saint Louis encephalitis virus (SLEV).
PRODUCT DETAILS – SAINT LOUIS ENCEPHALITIS VIRUS NS1 PROTEIN
- Recombinant Saint Louis Encephalitis virus NS1 protein (strain MS1-7, NCBI Accession Number: P09732.2) produced from HEK293 cells.
- Includes amino acids 760-1141 and a C-terminal His-tag.
- Greater than 95% purity and buffered in DPBS, pH7.4.
BACKGROUND
SLEV is within the Japanese encephalitis virus (JEV) complex along with other flaviviruses including Murray Valley encephalitis virus (MVEV) and West Nile virus (WNV).
The SLEV genome is ~11 kb, single-stranded, positive-sense RNA and encodes a polyprotein flanked by 5′ and 3′ untranslated regions. At the nucleotide and amino acid level, SLEV has much lower variation than other members of the Japanese encephalitis virus complex. The polyprotein is co- and post-translationally processed into the three structural proteins in the order, capsid (C), pre-membrane/membrane (prM/M) and envelope (E) and seven non-structural proteins: NS1, NS2A, NS2B, NS3, NS4A, NS4B and NS5.
NS1 involved in immune evasion, pathogenesis and viral replication. Following cleavage, it is targeted to three destinations; the viral replication cycle, the plasma membrane and the extracellular compartment. It is essential for viral replication where it is required for formation of the replication complex and recruitment of other non-structural proteins to the ER-derived membrane structures. It is also excreted as a hexameric lipoparticle that plays a role against host immune response. It binds to the host macrophages and dendritic cells, antagonizing the complement function and inhibiting signal transduction originating from Toll-like receptor 3 (TLR3).