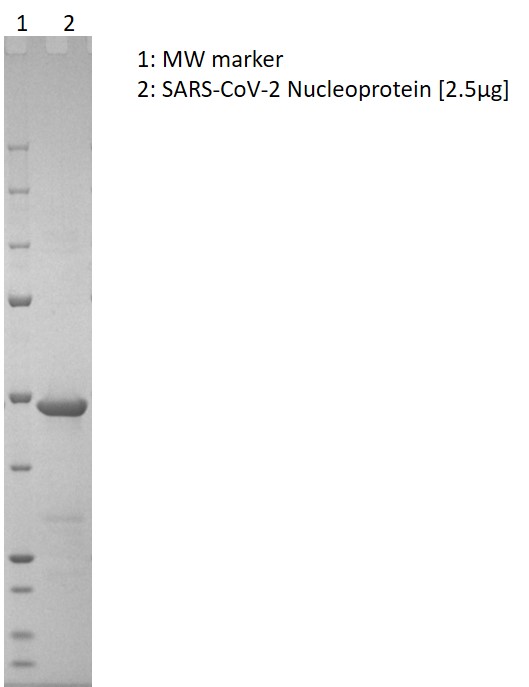
SDS-PAGE: Coomassie-stained SDS-PAGE showing purified SARS-CoV-2 nucleoprotein.
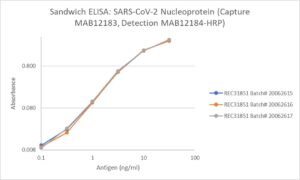
Sandwich ELISA: Minimal batch-to-batch variation in ELISA between different lots of REC31851. Greiner 762071 plates coated overnight with capture antibody MAB12183 (5µg/ml) in DPBS, 100µl/well at 23°C. Block 3X washes (300µl/well) 1% BSA in DPBS for 1.5 h, wash 2X300µl PBS + 0.1% Tween 20. Diluted antigen added in DPBS/1% BSA/0.1% Tween 20, 100µl/well, incubated shaken 3.5 h at 23°C. Washed 3X 300µl. Added detection antibody MAB12184-HRP (1µg/ml) in DPBS/1% BSA/0.1% Tween 20 100µl/well, incubated shaken 2 h at 23°C. Washed 6X 300µl. Added standard TMB 100µl/well, developed 10min, stopped & read at 450/620nm.
SARS-COV-2 NUCLEOPROTEIN, HIS-TAG (E. COLI)
SARS-CoV-2 Nucleoprotein (E. coli) is a recombinant antigen manufactured to high purity. SARS-CoV-2, previously known as the 2019 Novel Coronavirus (2019-nCoV), causes the pandemic COVID-19 disease.
PRODUCT DETAILS – SARS-COV-2 NUCLEOPROTEIN, HIS-TAG (E. COLI)
- Recombinant SARS-CoV-2 Nucleocapsid phosphoprotein (GenBank: MN908947, isolate Wuhan-Hu-1).
- Expressed and purified from E. coli as full-length nucleoprotein (amino acids 1-419), with a C-terminal 6xHis-tag.
- SARS-CoV-2 Nucleoprotein (E. coli) is purified to >90% purity and presented in 20mM sodium phosphate, 25mM potassium carbonate pH10.0, 150mM NaCl..
BACKGROUND
Coronaviruses (CoVs) are a diverse family of viruses which cause a variety of diseases in mammals and birds ranging from enteritis in cows and pigs and upper respiratory disease in chickens to potentially lethal human respiratory infections. Coronaviruses can cause a range of symptoms varying from mild symptoms such as the common cold to more serious respiratory illnesses. They primarily cause respiratory and enteric diseases in mammals and birds. Coronavirus symptoms include rhinorrhea, sneezing, cough, nasal obstruction, and bronchitis.
SARS-CoV-2 is a respiratory virus which causes coronavirus disease 2019 (COVID-19); it spreads primarily through contact with an infected person through respiratory droplets generated when a person coughs or sneezes, or through droplets of saliva or discharge from the nose. The incubation period is believed to range from 2-11 days. Infection with SARS-CoV-2 can cause mild symptoms including a runny nose, sore throat, cough, and fever. However, it can be more severe for some people and can lead to pneumonia or breathing difficulties. The elderly, and people with pre-existing medical conditions (such as, diabetes and heart disease) appear to be more vulnerable to becoming severely ill with the virus (WHO, 2020).
Coronaviruses encode five structural proteins in their genomes; namely, the Spike (S), Membrane (M), Envelope (E) glycoproteins, Hemagglutinin Esterase (HE) and Nucleoprotein (N), (Figure 2). All envelope proteins and N protein is present in all virions but HE is only present in some beta coronaviruses (Tok and Tatar, 2017). There are three main groups of coronaviruses: alpha, beta, and gamma. Nucleoproteins, also known as nucleocapsid proteins, are phosphoproteins that are capable of binding to helix and have flexible structure of viral genomic RNA. It plays an important role in virion structure, replication and transcription of coronaviruses, as it localizes in both the replication/ transcriptional region of the coronaviruses and the ERGIC region where the virus is collected. It packages the positive strand viral genome RNA into a helical ribonucleocapsid (RNP) and plays a fundamental role during virion assembly through its interactions with the viral genome and membrane protein M. It also plays an important role in enhancing the efficiency of subgenomic viral RNA transcription as well as viral replication. There is structural similarity between porcine reproductive and respiratory syndrome virus (PRRSV), a member of Arteriviridae, suggesting a common origin for Coronaviridae and Arteriviridae nucleoproteins (Yu et al., 2006).
REFERENCES
- Tok and Tatar (2017). Structures and Functions of Coronavirus Proteins: Molecular Modeling of Viral Nucleoprotein -.International Journal of Virology & Infectious Diseases. Vol 2, 1.
- Yu et al. (2006). Crystal Structure of the Severe Acute Respiratory Syndrome (SARS) Coronavirus Nucleocapsid Protein Dimerization Domain Reveals Evolutionary Linkage between Corona- and Arteriviridae. The Journal of Biological Chemistry, Vol. 281, No. 25, pp. 17134 –17139.
- Novel coronavirus (2019-nCoV), World health Organisation (WHO), 2020.