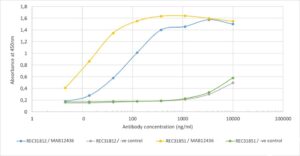
Antigen-down ELISA: Binding curve of Rabbit Anti SARS-CoV-2 Nucleoprotein Antibody (CR3018) to SARS-CoV-2 Nucleoproteins (REC31812 and REC31851). ELISA Plate coated with SARS-CoV-2 Nucleoproteins at a concentration of 5 µg/ml. A 3-fold serial dilution from 10,000 ng/ml was performed using MAB12436. For detection, a 1:4000 dilution of HRP-labelled anti-rabbit antibody was used. Rabbit anti-Fluorescein antibody was used as a negative control.
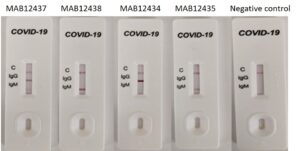
Rapid test: Antibodies used as positive control in COVID-19 IgM/IgG Rapid Test CE-IVD (UNCOV-40, Elabscience). The antibodies (from left to right) MAB12437 (CR3018, IgG1) and MAB12438 (CR3018, IgM), MAB12434 (CR3009, IgG1) and MAB12435 (CR3009, IgM), and negative control, were tested at 50 µg/ml diluted in human normal serum (SIGMA S1-100mL). Bands corresponding to IgG or IgM antibodies can be observed for CR3018 and CR3009, with no signal for the negative control.
Human IgG1 Anti-SARS-CoV-2 Nucleoprotein Antibody (CR3018)
Price range: £520.76 through £1,982.70 excl. VAT
Human IgG1 anti SARS-CoV-2 nucleoprotein antibody (clone CR3018) is a recombinant monoclonal antibody that recognizes and binds the amino acid residues between 11-19 of the N protein of SARS-CoV and SARS-CoV-2. SARS-CoV-2, previously known as the 2019 Novel Coronavirus (2019-nCoV), causes the pandemic COVID-19 disease.
HUMAN IgG1 ANTI-SARS-COV-2 NUCLEOPROTEIN ANTIBODY (CR3018)
Human anti SARS-CoV-2 Nucleoprotein antibody is a recombinant monoclonal antibody that recognizes the the SARS-CoV and SARS-CoV-2 nucleoprotein, the causative agent of COVID-19. Immunofluorescence staining was used to demonstrate binding of CR3018 to SARS-CoV infected Vero cells (van den Brink et al., 2005) and initial characterization of the antibody for binding to SARS-CoV-2 was carried out by ELISA.
PRODUCT DETAILS – HUMAN IgG1 ANTI-SARS-COV-2 NUCLEOPROTEIN
- Human anti SARS-CoV-2 Nucleoprotein antibody recognises and binds the amino acid residues between 11-19 of the N protein of SARS-CoV and SARS-CoV-2
- Isotype – Human IgG1, Kappa.
- The original antibody was generated by cloning the variable regions of the scFvs selected from phage display libraries into separate vectors for IgG1 heavy-chain and light-chain expression.
- Protein A affinity purified from culture supernatant and suitable for use in ELISA, LFA and IF.
- Competitive ELISA of this antibody with CR3009 suggests that both these antibodies bind different epitopes of the N protein of SARS-CoV. Thus, a combination of these two antibodies is suggested for virus capture assays.
BACKGROUND
The coronavirus nucleocapsid phosphoprotein (N, or nucleoprotein) is highly basic and binds the viral RNA as a dimeric entity into nucleocapsids which protect the viral genome, while also providing access for replication when required. The nucleoprotein packages the positive strand viral genome RNA into a helical ribonucleocapsid (RNP) and plays a fundamental role during virion assembly through its interactions with the viral genome and membrane protein M. It plays an important role in enhancing the efficiency of subgenomic viral RNA transcription as well as viral replication (Fan et al., 2005).
Antibodies directed against the nucleoprotein are useful tools for the development of diagnostics and the characteristics of both CR3009 and CR3018 could be useful in the development of a diagnostic test (Van Den Brink et al., 2005). A competition ELISA on immobilized SARS-CoV showed that binding of biotinylated IgG CR3009 and CR3018 was not or hardly affected in the presence of excess of unlabeled IgG CR3018 and CR3009, respectively. As expected binding of the biotinylated anti-N MAbs is blocked by their unlabeled counterparts but not by anti-S MAb CR3014. Therefore, IgG CR3009 and CR3018 do not compete with each other for binding to N protein and most likely recognize different epitopes. Pepscan analysis was used to map the epitopes of CR3009 and CR3018 IgG on the N protein. IgG CR3018 reacted with a continuous series of linear and looped peptides, starting with the sequences GPQSNQRSAPRITFG and PQSNQRSAPRITFGG, respectively, and both ending with the peptide RSAPRITFGGPTDST, thereby having the minimal sequence RSAPRITFG in common. This linear epitope is conserved in the N protein sequence of all published human SARS-CoV and animal SARS-CoV-like isolates but is absent in other members of the family of Coronaviridae. For IgG CR3009 the N protein Pepscan analysis did not reveal significant binding to any of the N protein-derived peptides, suggesting that it recognizes a nonlinear epitope.
REFERENCES
- Van Den Brink et al. (2005). Molecular and Biological Characterization of Human Monoclonal Antibodies Binding to the Spike and Nucleocapsid Proteins of Severe Acute Respiratory Syndrome Coronavirus. Journal of Virology (2005); Vol-79 (3); p. 1635–1644.
- Fan H, Ooi A, Tan YW, Wang S, Fang S, Liu DX, Lescar J. (2005). The nucleocapsid protein of coronavirus infectious bronchitis virus: crystal structure of its N-terminal domain and multimerization properties. Structure, 13 (12): 1859-68.