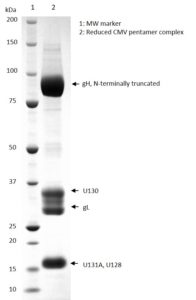
SDS-PAGE: Coomassie-stained SDS-PAGE showing Cytomegalovirus gH Pentamer Complex.
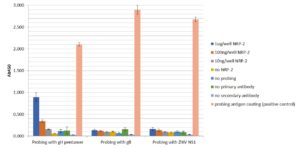
Direct ELISA: Plate was coated with 1000ng, 100ng, 10ng or 1ng of NRP-2 and probed with 500µg per well of NRP-2, gH pentamer or gB huFc-tag and then blocked for 1 h at room temperature. Plate was probed with 500µg per well of NRP-2, gH pentamer or gB huFc-tag for 2h at room temperature. For primary antibodies, sheep anti-gH pentamer serum was used at 1:2,000, mouse anti-His-tag antibody was used at 1:1,200 (for ZIKV-NS1), no primary antibody was used for gB. Secondary antibody STAR88P was used for gH pentamer at 1:2,000; STAR147P was used for gB huFc-tag detection via Fc-tag at 1:2,000 and Biorad 105300 was used to detect ZIKV-NS1 at 1:2,000. All secondary incubations were done for 45 min at room temperature. Detection was carried out using TMB Membrane substrate (KPL) for 40 min. 1M HCl was used to stop the reaction. Assay confimred that NRP-2 binds specifically to gH pentamer but not to gB or ZIKV NS1.
CYTOMEGALOVIRUS gH PENTAMER COMPLEX
Cytomegalovirus gH Pentamer Complex comprises five viral proteins (gH, gL, UL128, UL130, and UL131A). This complex has been shown to bind our recombinant NRP-2 protein in ELISA.
PRODUCT DETAILS – CYTOMEGALOVIRUS gH PENTAMER COMPLEX
- Recombinant human cytomegalovirus pentamer protein complex (strain VR1814), consisting of proteins UL75 (gH), UL115 (gL), UL128, UL130 and UL131A.
- The gH protein contains a C-terminal His-tag and is produced recombinantly in HEK293 cells.
- Presented in Dulbecco’s PBS, 0.2µm filter sterilised.
- Purified protein binds NRP-2 receptor in ELISA.
- NCBI Accession Code: ACZ79986.
BACKGROUND
hCMV can cause serious morbidity/mortality in immune compromised individuals such as transplant recipients and HIV patients, and congenital hCMV infection can lead to birth defects.
The Pentamer Complex is expressed on all wildtype hCMV strains but is notably missing from laboratory strains such as AD169. (Freed et al.). This has placed severe limitations on vaccine development. Receptor-mediated viral entry into endothelial cells requires a functional gH pentameric complex, and this complex is one of the primary targets for antiviral antibodies in infected individuals. NAC’s new recombinant hCMV gH complex will be key to enabling essential basic and applied research. In addition, the gH complex is expected to provide the basis for a new generation of IVD tests.
The native-like Pentamer Complex is produced in human cells to ensure that all proteins are properly folded and possess their native glycosylation pattern. The presence of all five proteins in the purified complex is confirmed by mass spectrometry. The Pentamer Complex is offered in ready to use liquid format and is available 100ug and 1mg vials size with the opportunity to purchase bulk quantities.
NAC offers two further antigens in its CMV range: a native high purity antigen which demonstrates a high affinity for both IgG and IgM, ideal for antibody captures assays, and a standard lysate more suited as a serological control.
Cytomegalovirus gH pentamer complex is sold as a catalogue item for research use and for use in in vitro diagnostic assay/kits only and as such is without the need for prior agreement with NAC. If the product is intended to be used as a pharmaceutical or as a means to produce a pharmaceutical (including use as an immunogen and for clinical trials), then this must be under prior contractual agreement with NAC.
References