SDS-PAGE: Reducing SDS-PAGE gel showing purified Chikungunya E2 envelope protein (Human Fc tag) running with an expected MW of approximately 70-75kDa.
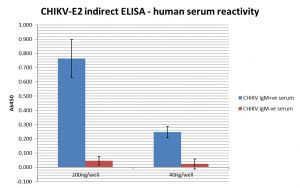
ELISA: ELISA assay of purified Chikungunya virus E2 envelope protein (Human Fc tag), showing binding of Chikungunya IgM +ve patient sera.
CHIKUNGUNYA VIRUS E2 ENVELOPE PROTEIN (HEK293), HUMAN FC-TAG
Chikungunya Virus E2 Envelope protein is a unique product that has been developed in response to the need for high purity, properly assembled and glycosylated Chikungunya virus antigens for use in the development of Chikungunya virus diagnostics and in vaccine development and R&D (including use as an immunogen).
Chikungunya Virus E2 Envelope protein is produced in human cell lines using state-of-the-art expression techniques. This protein is prepared with a human IgG1 Fc-tag, and is purified by Protein A affinity chromatography. We also offer the protein with a mouse IgG2a Fc-tag for those customers where a human Fc-tag may interfere with their assays.
PRODUCT DETAILS – CHIKUNGUNYA VIRUS E2 ENVELOPE PROTEIN (HEK293), HUMAN FC-TAG
- Recombinant Chikungunya Virus E2 Envelope Protein (amino acids 326-693 of the polyprotein, NCBI Accession Number: Q5XXP3.1).
- xpressed in HEK293 cells with a C-terminus human IgG1 Fc tag.
- Ideal for use in detection of Chikungunya virus via IgG/IgM ELISA or rapid test device.
BACKGROUND
It has been reported that E2 protein may offer higher sensitivity in serological assays when compared to E1 protein (Cho et al, 2008). Chikungunya virus is a member of the genus Alphavirus in the family Togaviridae. Chikungunya fever is a mosquito-borne disease first identified in Tanzania in 1953. Since 2004 there have been extensive outbreaks in Africa and Asia, and in 2013 the first cases were identified in the Caribbean, and by September 2014 more than 650,000 cases had been reported in the Americas. Chikungunya fever usually starts 2–4 days after chikungunya virus infection, with clinical symptoms including high fever, rash, headache, vomiting, myalgia, and severe joint pain.
Because the virus is transmitted by the same mosquitos that transmit Dengue Virus and Zika Virus, and the clinical symptoms of infection with the viruses are also similar, it is important to be able to distinguish diagnostically between the different infections. Because the use of aspirin and other NSAIDs can increase the risk of haemmorhage in Dengue fever, patients should be treated as having Dengue fever until a diagnosis of Dengue fever has been excluded.