RABBIT ANTI-BORRELIA BURGDORFERI SENSU STRICTO (B31) OSPB ANTIBODY
This is a polyclonal antibody, prepared against OspB from the spirochete B. burgdorferi, for use in ELISA and western blotting applications. Strain B31 is the type strain (ATCC 35210) for this organism and was derived by limited dilutional cloning from the original Lyme-disease tick isolate obtained by A. Barbour (Johnson, et al., 1984).
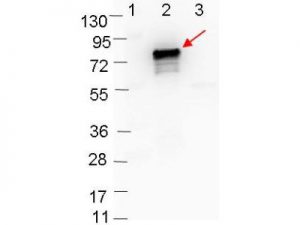
PRODUCT DETAILS – RABBIT ANTI-BORRELIA BURGDORFERI SENSU STRICTO (B31) OSPB ANTIBODY
- Rabbit anti-B. burgdorferi sensu stricto OspB polyclonal IgG antibody (strain B31).
- Greater than 95% purity by SDS-PAGE and buffered in 0.02 M Potassium Phosphate, 0.15 M Sodium Chloride, pH 7.2.
BACKGROUND
Many of the borrelial surface antigens are lipid-modified proteins (i.e. lipoproteins), although a number of these surface-exposed lipoproteins (OspA, OspB, and OspC) are not found exclusively on the surface of the organism. These lipoproteins are also detected in the periplasm of the organism and can be shuttled to and from the borrelial surface at different points during infection (Haake, 2000).
The interface between B. burgdorferi and its human host is its outer surface (and therefore proteins localized to the outer membrane) play an important role in dissemination, virulence, tissue tropism, and immune evasion. Antibodies directed against outer surface proteins have also been shown to protect animals and humans from infection with B. burgdorferi (Kenedy, et al., 2012). OspA (31 kDa) and OspB (34 kDa) proteins show a high degree of sequence similarity, are encoded by a 49-kb plasmid, share a common promoter and are co-ordinately transcribed (Liang, et al., 2004).
All three Osp proteins (OspA, OspB and OspD) are expressed by B. burgdorferi residing in the gut of unfed ticks, suggesting that they promote the persistence of the spirochete in ticks between blood meals. OspB has a contributing role in the adherence of B. burgdorferi to the tick gut. The C terminus of OspB is important for eliciting a protective immune response to OspB. B. burgdorferi has the ability to vary its surface proteins in response to immune attack (Neelakanta, et al., 2007).
REFERENCES
- Haake, D. A., 2000. Spirochaetal lipoproteins and pathogenesis. Microbiology , 147(7), pp. 1491-1504.
- Johnson, R.C., et al. 1984. Borrelia burgdorferi sp. nov.: etiologic agent of Lyme disease. Int J Syst Bacteriol, 34, pp. 496–497.
- Kenedy, M. R., Lenhart, T. R. & Akins, D. R., 2012. The Role of Borrelia burgdorferi Outer Surface Proteins. FEMS Immunol Med Microbiol., 66(1), pp. 1-19.
- Liang, F. T., Caimano, M. J., Radolf, J. D. & Fikrig, E., 2004. Borrelia burgdorferi outer surface protein (osp) B expression independent of ospA. Microb Pathog., 37(1), pp. 35-40.
- Neelakanta, G. et al., 2007. Outer surface protein B is critical for Borrelia burgdorferi adherence and survival within Ixodes ticks. PLoS Pathog, 3(3).